The Unseen Architects: How Cryptic Pockets Are Revolutionizing Drug Discovery
For decades, drug discovery has largely focused on targeting well-defined binding sites on proteins – the so-called "druggable" pockets. However, a vast number of disease-related proteins remain stubbornly "undruggable," lacking readily apparent sites for therapeutic intervention. This challenge has spurred scientists to look beyond the obvious, leading to a groundbreaking realization: proteins are not static structures, but dynamic shape-shifters that can reveal transient, hidden binding sites known as "cryptic pockets." These elusive cavities are now emerging as a transformative frontier, promising to unlock new avenues for developing life-changing medications and fundamentally changing our understanding of how drug candidates exert their impact on pocket dynamics and overall protein function.
The concept of cryptic pockets challenges traditional paradigms, suggesting that a protein's true therapeutic potential might lie in its less stable, fleeting conformations. By understanding these dynamic structural changes and how to exploit them, researchers are gaining an unprecedented ability to target previously inaccessible proteins, modulate their activity in novel ways, and design drugs with enhanced specificity and efficacy. This paradigm shift holds immense promise for addressing a wide array of diseases, from infectious diseases to cancer and neurodegenerative disorders.
Unveiling the Hidden World of Cryptic Pockets
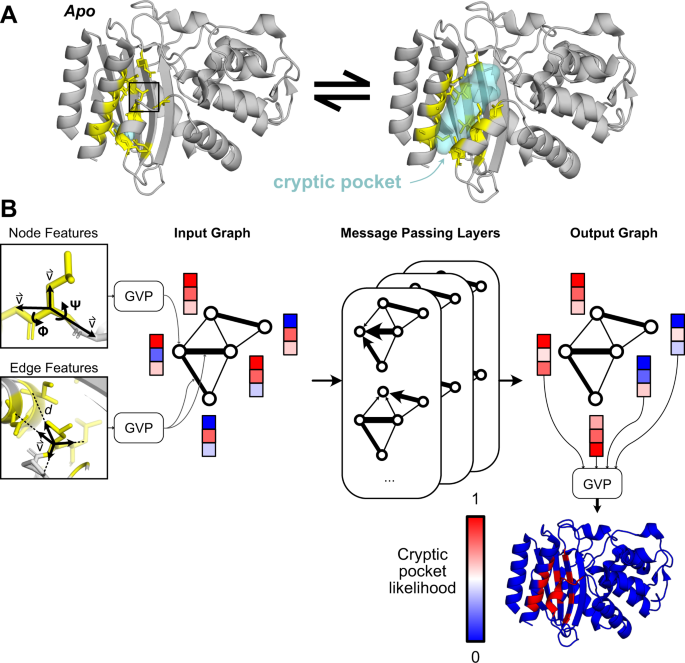
At the heart of this revolution is the understanding that proteins exist in a constant state of flux, sampling various conformations. While crystallographic structures often capture a single, dominant form, proteins frequently adopt a multitude of "excited states" – transient, low-population conformers that are crucial for their biological function. Cryptic pockets are precisely these hidden cavities that are absent in the most stable, ligand-free structures but become exposed or form de novo as the protein undergoes these dynamic conformational changes.
Identifying and characterizing these ephemeral pockets presents a significant scientific hurdle. Their transient nature makes them difficult to observe using conventional structural biology techniques like X-ray crystallography or cryo-electron microscopy, which typically favor stable states. This challenge necessitates the use of advanced computational methods, such as molecular dynamics simulations, combined with sophisticated experimental techniques like nuclear magnetic resonance (NMR) spectroscopy and kinetic assays, to catch these fleeting moments of pocket formation.
The importance of cryptic pockets lies in several key aspects:
- Accessing "Undruggable" Targets: They offer novel binding sites on proteins previously deemed untargetable due to the absence of conventional pockets.
- Novel Modulatory Mechanisms: Drugs can be designed to bind to these cryptic sites, allosterically modulating protein function (enhancing or inhibiting it) rather than competing with the natural substrate at the active site. This can lead to greater specificity and fewer off-target effects.
- Enhancing Protein Function: Unlike most drugs that aim to inhibit protein function, some cryptic pockets can be targeted to *increase* beneficial protein activity, opening up entirely new therapeutic strategies.
The Beta-Lactamase Case Study: A Glimpse into Functional Impact
A seminal study published in PNAS regarding β-lactamase enzymes beautifully illustrates the functional significance of cryptic pockets. β-lactamases are bacterial enzymes that confer resistance to β-lactam antibiotics by hydrolyzing the antibiotic molecule. The study specifically investigated the Ω-loop cryptic pocket within these enzymes, a site not readily apparent in their standard structures.
Researchers employed a multi-faceted approach, combining thiol-labeling and kinetic assays with NMR and molecular dynamics simulations, to explore the dynamics and functional consequences of this hidden pocket. Their findings were striking: an open Ω-loop cryptic pocket population was found to be beneficial for the hydrolysis of the substrate benzylpenicillin. This indicates a direct and positive impact on pocket function, where the transient opening of this specific pocket enhances the enzyme's catalytic efficiency.
This research provided crucial evidence that excited states and their associated cryptic pockets do not merely arise from thermal fluctuations that detract from function. Instead, they can actively *enhance* protein function, directly influencing the enzyme's ability to perform its biological role. The presence of this Ω-loop pocket, even if not completely conserved across all β-lactamase homologs, correlated with higher activity against benzylpenicillin, particularly in variants exhibiting a greater open pocket population. This underscores the critical impact on pocket conformation on overall enzyme performance.
Implications for Next-Generation Drug Design
The discovery and characterization of cryptic pockets are poised to have a profound impact on pocket-centric drug discovery, fundamentally reshaping how we approach pharmaceutical development. No longer are drug designers confined to static representations of proteins. Instead, they can embrace the dynamic nature of these biomolecules, searching for transient, targetable sites that emerge during conformational changes.
This dynamic view opens the door to:
- Rational Design for "Undruggable" Targets: By identifying cryptic pockets on previously intractable proteins (e.g., certain transcription factors, adaptor proteins, or intrinsically disordered proteins), scientists can design small molecules that stabilize these transient open states or bind exclusively to them.
- Allosteric Modulators: Drugs can be developed to bind to cryptic pockets located away from the active site, inducing conformational changes that either enhance or inhibit the protein's function. This allosteric modulation offers advantages in terms of specificity and reduced toxicity compared to active-site inhibitors.
- Exploiting Functional Enhancement: For certain diseases, therapeutic intervention might involve *boosting* a protein's activity. Cryptic pockets provide a mechanism to achieve this, moving beyond the traditional inhibition-focused drug discovery.
- Overcoming Drug Resistance: In cases like β-lactamase, understanding cryptic pockets could lead to the development of new inhibitors that target these dynamic sites, potentially circumventing existing resistance mechanisms.
Advanced computational methods, including artificial intelligence and machine learning, are becoming indispensable tools for predicting the existence and dynamics of cryptic pockets, screening vast chemical libraries for potential binders, and optimizing lead compounds. This synergistic approach between experimental validation and computational power accelerates the drug discovery pipeline significantly, promising more effective and targeted therapies.
Beyond Proteins: The Universal Importance of Pocket Dynamics in Health
While the focus on cryptic pockets primarily revolves around protein dynamics and their role in drug discovery, the broader concept of understanding "pockets" in biological systems and the factors that influence their state is universally critical for health and disease. Just as a transient cryptic pocket can determine a protein's function, other biological pockets, such as those found in periodontal health, can significantly impact overall well-being. The principles of understanding how a system's environment and specific factors can alter these cavities, leading to either beneficial or detrimental outcomes, resonate across different biological scales.
For instance, in the context of oral health, periodontal pockets – the spaces between teeth and gums – are critical indicators of disease. The closure of these pockets after treatment is a primary goal in managing periodontitis. Research has clearly demonstrated how external factors, much like conformational changes in proteins, can have a profound impact on pocket health and closure rates. Specifically, studies have shown that factors such as smoking can severely impede the success of nonsurgical periodontal treatment, leading to poorer pocket closure outcomes even in the absence of residual inflammation. This highlights the importance of patient lifestyle choices in influencing biological pocket dynamics and therapeutic success.
Understanding the interplay of factors that influence any biological "pocket," be it a protein binding site or a periodontal space, is key to achieving optimal clinical outcomes. For those interested in how environmental factors impact on pocket health in other biological contexts, such as dental care, exploring the significance of Smoking Cessation Boosts Periodontal Pocket Closure Success and understanding Periodontal Pocket Closure: Factors Beyond Nonsurgical Treatment can provide further insights into the diverse mechanisms at play.
Navigating the Future: Challenges and Opportunities
The journey to fully harness the potential of cryptic pockets is not without its challenges. The transient nature of these pockets means their identification and characterization remain technically demanding and computationally intensive. Moreover, designing molecules that selectively bind to a specific cryptic pocket without affecting other parts of the protein or off-target proteins requires exquisite precision. The complexity of protein dynamics demands ever more sophisticated experimental techniques and advanced computational modeling to accurately predict and validate cryptic pocket formation and ligand binding.
Despite these hurdles, the opportunities are immense. As our understanding of protein dynamics deepens and experimental and computational tools become more powerful, cryptic pockets will undoubtedly become a cornerstone of future drug discovery efforts. They represent a fundamental shift in perspective, moving from static lock-and-key models to a dynamic induced-fit paradigm where proteins actively reveal their therapeutic vulnerabilities. This exciting frontier promises to unlock a new generation of drugs, offering hope for conditions that are currently untreatable and significantly improving patient outcomes worldwide.